 The sum of all bond energies in such a molecule is equal to the standard enthalpy change for the endothermic reaction that breaks all the bonds in the molecule. Molecules with three or more atoms have two or more bonds. The bond energy for a diatomic molecule, D X–Y, is defined as the standard enthalpy change for the endothermic reaction: The energy required to break a specific covalent bond in one mole of gaseous molecules is called the bond energy or the bond dissociation energy. The stronger a bond, the greater the energy required to break it. Separating any pair of bonded atoms requires energy (see Figure 7.4). We measure the strength of a covalent bond by the energy required to break it, that is, the energy necessary to separate the bonded atoms. Stable molecules exist because covalent bonds hold the atoms together. In this section, you will learn about the bond strength of covalent bonds, and then compare that to the strength of ionic bonds, which is related to the lattice energy of a compound. Use average covalent bond energies to estimate enthalpies of reactionĪ bond’s strength describes how strongly each atom is joined to another atom, and therefore how much energy is required to break the bond between the two atoms.Use the Born-Haber cycle to compute lattice energies for ionic compounds.Describe the energetics of covalent and ionic bond formation and breakage. 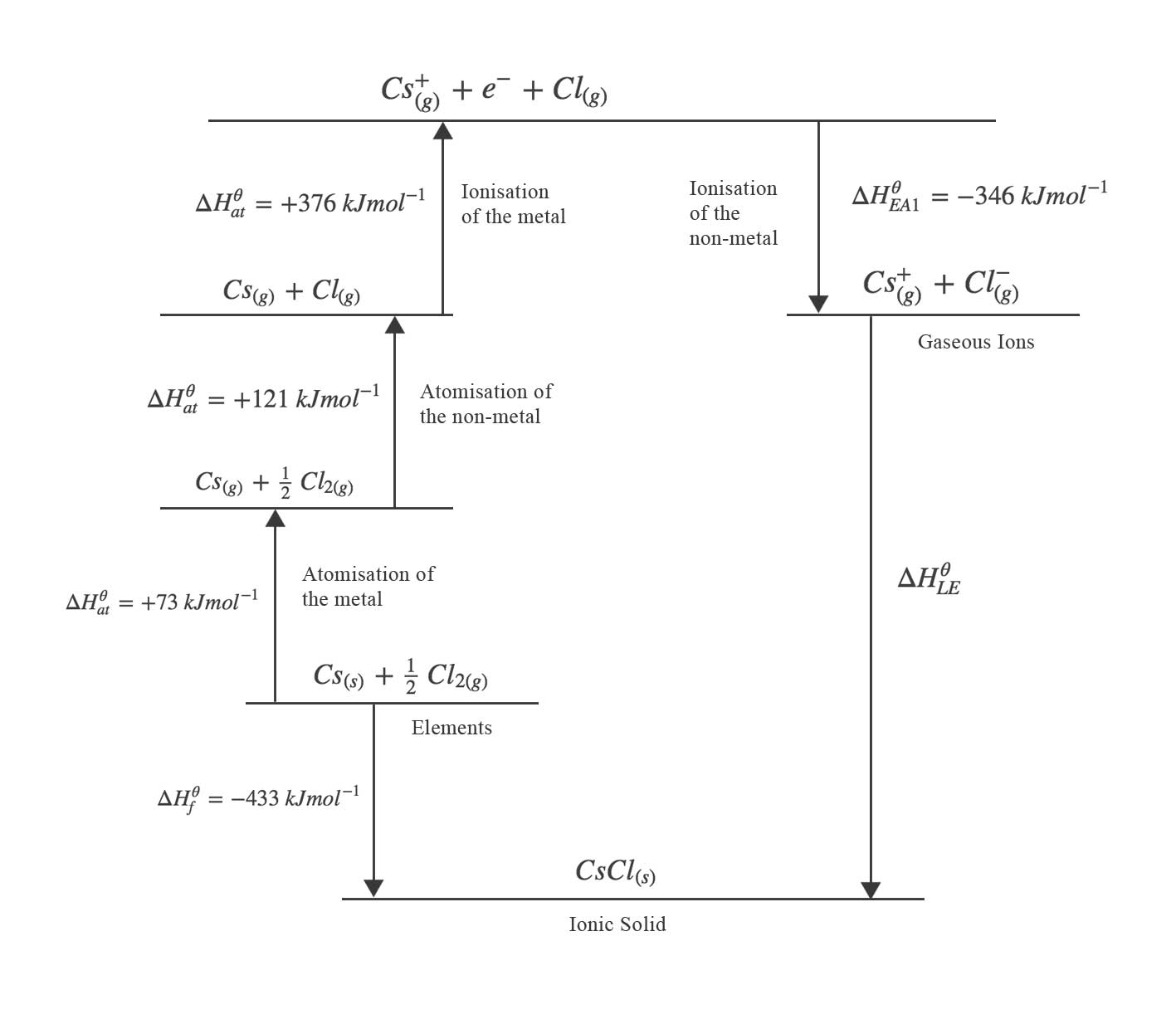 
By the end of this section, you will be able to: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
